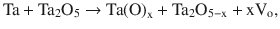1. Major Degradation Mechanisms
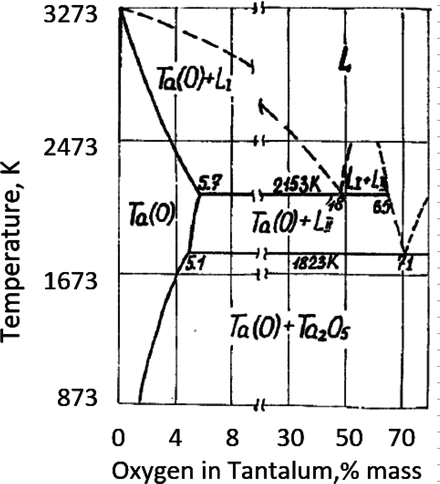
The basic bilayer of all types of tantalum capacitors, tantalum anode, and anodic oxide film of tantalum as a dielectric is not a thermodynamically stable system. This is demonstrated by the tantalum-oxygen equilibrium diagram that does not contain two-phase equilibrium areas for pure tantalum and tantalum pentoxide (Fig. ]. Tantalum capacitors with thinner dielectrics are more sensitive to oxygen migration since the thickness of the depleted with oxygen portion of dielectric near the Ta-Ta2O5 interface is comparable with the total thickness of the dielectric.
Fig. 1.1
Tantalum-oxygen equilibrium diagram []
Another reason for the thermodynamic instability of tantalum capacitors is the amorphous structure of the anodic oxide film of tantalum formed on crystalline tantalum. The image of the Ta-Ta2O5 bilayer on Fig. ].
Fig. 1.2
TEM image of the amorphous anodic oxide film formed on crystalline tantalum (the white spots represent individual atoms)
One can see from Fig. that atoms in the anodic oxide film of tantalum are disordered and do not have any preferable orientation, which is inherent to amorphous structures, while atoms in the tantalum metal are ordered in a crystalline lattice. This amorphous structure of the anodic oxide film of tantalum employed as a dielectric in tantalum capacitors is of paramount importance to performance and reliability of these capacitors. Typically, the dielectrics in the tantalum capacitors are very thin (from 0.02 m to 0.6 m depending on the formation voltage). As a result of the low thickness, the electrical field in the dielectric film at rated voltage is on the order of several million V/cm. The disordered arrangement of atoms in the amorphous dielectric results in a high density of electron traps for the electric current carriers. Due to these traps, the mobility of electric current carriers stays low in the high electrical field, preventing accumulation of critical energy that otherwise might cause an electric avalanche and the breakdown of the dielectric.
On the other hand, the disordered structure of the amorphous dielectrics increases the internal energy in the dielectric, making these dielectrics thermodynamically unstable. Amorphous dielectrics trend to ordering and crystallization spontaneously to reduce their internal energy. Growth of crystalline inclusions in amorphous matrix of the anodic oxide film induces mechanical stress in the film due to a difference between specific volume of the amorphous and crystalline phases. Mechanical stress associated with field-induced crystallization was detected experimentally using in situ measurements on anodic oxide film of tantalum deposited on quartz substrate []. Eventually this mechanical stress results in a disruption of the dielectric and, thereby, in the failure of the capacitor. The rate of crystallization process is a principal factor governing the performance and reliability of tantalum capacitors.
The rate of the crystallization process in anodic oxide films on tantalum depends on many factors such as purity and morphology of the anode surface, type and temperature of formation electrolyte, conditions of post-formation thermal treatment, etc. In all cases, the crystallization rate increases with increasing thickness of the anodic oxide film. The effect of the dielectric thickness on the rate of crystallization can be explained by contributions to the internal energy of the dielectric by the bulk of the anodic oxide film and the interface between the anodic oxide film and anode. An amorphous film doesnt need misfit dislocations on the interface with the crystalline anode in order to adjust its structure to the crystalline lattice of the anode. Since these misfit dislocations increase internal energy, the amorphous film adjacent to the crystalline substrate allows lower internal energy than a similar crystalline film would be. This helps stabilize the amorphous state of the dielectric. On the other hand, the disordering of atoms in the bulk of the amorphous dielectric increases internal energy of the dielectric. The balance in internal energy of the dielectric and therefore the balance between the tendency of the dielectric to remain amorphous or to crystallize depend on the ratio between the surface and bulk of the dielectric. The greater is the thickness of the amorphous dielectric, the larger is the contribution of the bulk to the internal energy that makes thicker amorphous dielectrics more prone to crystallization.
Despite the inherent thermodynamic instability, tantalum capacitors can be manufactured with exceptional stability and reliability. Such high stability and reliability can be achieved because kinetics of the degradation processes, oxygen migration and crystallization, can be reduced by technological means to such a negligible level that no degradation takes place during any practical duration of the testing and field application, even at the highest possible operation temperatures and voltages. Oxygen migration and crystallization processes also take place in niobium-based capacitors, causing parametric and catastrophic failures of these capacitors. The pace of the degradation processes in niobium-based capacitors at given dielectric thickness, temperature, and applied voltage is significantly higher than that in tantalum capacitors with the same dielectric thickness, temperature, and voltage. Nevertheless, improvements in the stability and reliability of niobium-based capacitors have been also achieved by the technological means slowing down degradation processes in these capacitors.
1.1 Oxygen Migration
Comprehensive investigation of oxygen migration in tantalum capacitors was conducted during the early stages of manufacturing of solid MnO2 tantalum capacitors []. The manufacturing includes multiple pyrolysis of liquid manganese nitrite impregnating porous tantalum anodes to produce a solid MnO2 cathode. Thermal treatment associated with this process, typically performed in the range of temperatures 250350 C, resulted in an increase in DCL, dissipation factor (DF), and capacitance, as well as in a large capacitance dependence on bias, frequency, and temperature. Only at very low temperatures, there were practically no capacitance dependence on bias and frequency as a result of the thermal treatment.
To explain the effects of the thermal treatment on capacitance behavior of tantalum capacitors, a conductivity profile model was developed based on a gradient of oxygen vacancies in the oxide dielectric []. The conductivity profile develops during the thermal treatment as a result of oxygen migration from the tantalum oxide to the tantalum, leaving oxygen vacancies in the oxide dielectric. This transformation can be presented as:
where Ta(O)x is a solid solution of oxygen in tantalum and x is a concentration of oxygen in tantalum and concentration of oxygen vacancies Vo in tantalum oxide. When the oxygen deficiency reaches the oxide-air interface, oxygen begins to diffuse through the oxide ultimately producing a gradient of oxygen vacancies each with two trapped electrons. This oxygen vacancy gradient is associated with a gradient of conductivity since electrons trapped in the oxygen vacancies can be emitted under applied voltage, increasing conductivity of the oxide dielectric. The point of highest oxygen vacancy concentration and associated conductivity is located at the metal-oxide interface, after which the level of conductivity decreases exponentially across the dielectric.




![Freeman - Pro design patterns in Swift: [learn how to apply classic design patterns to iOS app development using Swift]](/uploads/posts/book/201359/thumbs/freeman-pro-design-patterns-in-swift-learn-how.jpg)