Junctional Melanocytic Hyperplasia
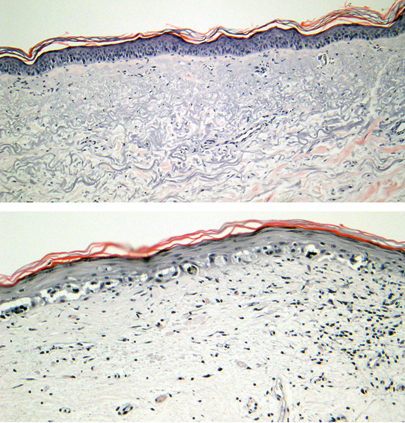
The term junctional melanocytic hyperplasia (JMH) refers to the proliferation of melanocytes along the dermoepidermal junction that is the basal layer where normal melanocytes reside. JMH is not a clinical diagnosis but a histological term, just as epidermal hyperplasia or nodular lymphocytic infiltrate . JMH generally refers to the proliferation of single melanocytes rather than nests of melanocytes, and is characteristic of lentigo simplex ; hence, the term lentiginous is often used to describe this finding.
Some pathologists make the diagnosis of lentiginous nevus for a junctional nevus with prominent lentiginous (or single cell) melanocytic hyperplasia. Others use the term jentigo to describe such proliferation, implying a lesion with features of both lentigo and early junctional nevus. Clinically, these lesions are usually very dark but small. Biologically, they represent the early stages of a melanocytic nevus and are common in childhood and youth. It is their dark and sometimes black color that causes concern by the patient, leading to their excision. Both, lentigo simplex and lentiginous junctional nevus are easily diagnosed histologically .
So What Is Atypical JMH?
Many times, the pathology report for a biopsy specimen in which the clinician suspected solar lentigo versus lentigo maligna carries the diagnosis of atypical junctional melanocytic hyperplasia. This is a histological conceptual diagnosis and not a clinical diagnosis, at least not for the time being.
In order to understand this concept, it is important to go to the other end of the spectrum of junctional melanocytic proliferations, namely melanoma in situ.
The diagnosis of melanoma in situ is made with ease in biopsy specimens that reveal diffuse proliferation of uniformly atypical melanocytes (melanoma cells), at least in the basal layer with or without involvement of the outer epidermal layers. In most cases of the lentigo maligna type of melanoma in situ, the proliferation is limited to the basal layer; while in most cases of the superficial spreading type of melanoma in situ, the proliferation is throughout all layers of the epidermis.
The histological diagnosis of atypical JMH is generally made when the proliferation is more than simple JMH of a junctional or dysplastic nevus, but short of that in melanoma in situ.
So what is a practitioner to do?
The answer to this question requires a discussion of known causes with a report of atypical JMH.
Causes of Atypical JMH
The first is severe chronic sun damage . Biopsies of the skin of the face for other lesions in a patient with severe chronic sun damage often reveal junctional melanocyte proliferation , which is a common response of junctional melanocytes to chronic ultraviolet damage. This is commonly observed over fibrous papules, dermal nevi, and other face lesions, occasionally to such intensity as to overshadow the dermal pathology especially in incomplete superficial biopsy specimens. A pathologist may decide to report on it or not. Biopsy of normal-appearing adjacent skin would most likely reveal similar findings.
The second cause of atypical JMH is trauma , characteristically by a recent surgical procedure . Re-excision specimens of recently biopsied neoplasms often reveal junctional melanocytic proliferation that was not present in the original recent biopsy specimen and is believed to be secondary to the process of wound healing. This is often easy to identify over a new scar and is rarely reported on by the pathologist.
The third is lentigo maligna , which is sun-induced melanoma in situ with limited invasive potential. Lentigo maligna has displaced superficial spreading melanoma as the most common type of melanoma. Practitioners are well aware of the clinical appearance of a large, irregularly pigmented and irregularly bordered patch on chronically sun-exposed skin, especially the face. For the accurate histological diagnosis of lentigo maligna, an adequate-sized biopsy or multiple biopsies are often required. This is due to the fact that unlike other types of melanoma, the degree of proliferation is somewhat variable within the lesion. Where the proliferation is mild, the diagnosis may be missed and in the absence of characteristic findings of another identifiable melanocytic neoplasm, a pathologist may simply make the diagnosis of atypical JMH .
In the authors experience, a second or larger biopsy specimen of a lesion clinically suspected to be lentigo maligna often reveals characteristic findings that confirm the diagnosis. Hence, it is possible to avoid receiving the diagnosis of atypical JMH caused primarily by a small biopsy specimen of lentigo maligna if multiple small biopsies or preferably a wide biopsy is submitted initially.
Since patients with lentigo maligna are highly likely to have a background, or so-called field effect, of significant JMH due to severe sun damage, how is a surgeon or pathologist to determine clear margins on a lentigo maligna excision commonly performed by the staged Mohs technique?
This question is asked to pathologists on a regular basis. Lentigo maligna is characterized by almost continuous proliferation of single melanocytes in the basal layer compared to the ratio of one melanocyte for every six to eight basal cells in non-sun-exposed skin, and a larger ratio in sun-damaged skin. As successive layers of peripheral skin are removed by the Mohs technique, at what ratio should a pathologist be comfortable that the lentigo maligna has been completely excised?
There are no guidelines that help answer this question. Both, pathologists and dermatologic surgeons vary in their stringency of defining the borders of lentigo maligna , possibly explaining the occasional recurrence of lentigo maligna following Mohs surgical excision. Some pathologists use immunohistochemical markers to identify proliferating melanocytes.
It is reasonable to say that lentigo maligna should be excised to its border with background skin which, as mentioned above, highly likely has some degree of JMH due to chronic sun damage. But how is a pathologist to know the degree of this background melanocytic hyperplasia? In the authors experience, a 23 mm biopsy of normal-appearing skin of the contralateral side has been extremely helpful in identifying the degree of background melanocytic hyperplasia, especially in cases where the lentigo maligna does not seem to end after several layers of excision.